Biochemical compounds- Mackenzie
Curriculum Expectations
B3.2 describes the structure of important biochemical compounds, including carbohydrates, proteins, lipids and nucleic acids and explains their function within cells.
B3.3 identify common functional groups within biological molecules and explain how they contribute to they contribute to the function of each molecule.
B2.3 construct and draw three-dimensional molecules models of important biochemical compounds, including carbohydrates, proteins, lipids and nucleic acids.
B2.4 conduct biological tests to identify biochemical compounds found in various food samples and compare the biochemical compounds found in each food to those found in each food to those found in the others.
B3.3 identify common functional groups within biological molecules and explain how they contribute to they contribute to the function of each molecule.
B2.3 construct and draw three-dimensional molecules models of important biochemical compounds, including carbohydrates, proteins, lipids and nucleic acids.
B2.4 conduct biological tests to identify biochemical compounds found in various food samples and compare the biochemical compounds found in each food to those found in each food to those found in the others.
Learning Goals
Students will:
1. Describe the function of the four types of biochemical compounds and explain their role in the cell.
2. Identify the functional groups of the four biochemical compounds and how they contribute to their functionality within the cell.
3. Construct 3-D representations of these molecules, which model the importance of these compounds
4. Identify the biochemical compound found in food samples using various biological tests.
1. Describe the function of the four types of biochemical compounds and explain their role in the cell.
2. Identify the functional groups of the four biochemical compounds and how they contribute to their functionality within the cell.
3. Construct 3-D representations of these molecules, which model the importance of these compounds
4. Identify the biochemical compound found in food samples using various biological tests.
All biochemical compounds contain carbon. Biochemical compounds contain other element such as hydrogen oxygen nitrogen phosphorous and a few other elements. It is the interactions and structural arrangement that determined the reactions of these biochemical compounds. Biochemical compounds are known as macromolecules. Theses macromolecules are made of single subunits, known as monomers. These monomers are linked together through dehydration reactions that form covalent bonds Biochemical compounds are organized into four categories of macromolecules; carbohydrates, proteins, lipids and nucleic acids.
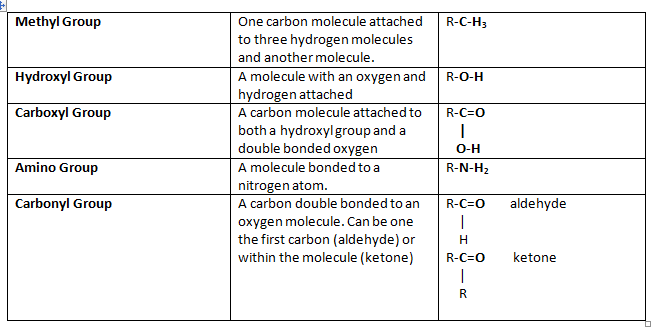
functional groups
- All compounds contain different functional groups
- Determine how a molecule bond and interact with one another
- Specific arrangement of atoms hold particular chemical property
- Determine how a molecule bond and interact with one another
- Specific arrangement of atoms hold particular chemical property
carbohydrates

Characteristics
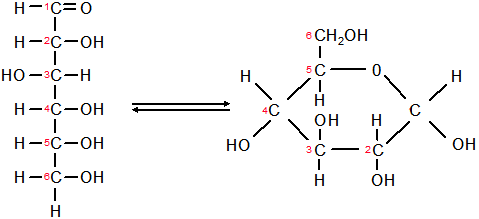
- Formed from monomers called monosaccharaides (glucose and fructose)
- Glucose in a monomer or monosaccharide. Two glucose molecules can bind together, through a dehydration reaction, to form a disaccharide, known as maltose. When three or more monomers of glucose bind together, they form a polysaccharide.
- Basic chemical formula is Cn(H2O)n
Use
- Provide energy by breaking bonds
- Storage product for energy
- Cell structure Ex. Cellulose used to form the plant cell wall.
Functional Groups
- Carbonyl group (aldehyde or ketone) and many hydroxyl groups, when straight chain
- Carbonyl group can react to form a ring (glucose)
- Formed from monomers called monosaccharaides (glucose and fructose)
- Glucose in a monomer or monosaccharide. Two glucose molecules can bind together, through a dehydration reaction, to form a disaccharide, known as maltose. When three or more monomers of glucose bind together, they form a polysaccharide.
- Basic chemical formula is Cn(H2O)n
Use
- Provide energy by breaking bonds
- Storage product for energy
- Cell structure Ex. Cellulose used to form the plant cell wall.
Functional Groups
- Carbonyl group (aldehyde or ketone) and many hydroxyl groups, when straight chain
- Carbonyl group can react to form a ring (glucose)
Lipids

Characteristics
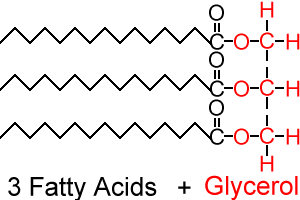
- Built of long fatty acid chains. These are long chains of hydrocarbon with a carboxyl group at the end.
- Example is triglycerides, which contain one glycerol and three fatty acid chains
Use
- Energy storage
- plasma membrane of cells
- internal signalling (hormones)
Functional Groups
- Carboxyl group react with glycerol to form triglyceride
- Built of long fatty acid chains. These are long chains of hydrocarbon with a carboxyl group at the end.
- Example is triglycerides, which contain one glycerol and three fatty acid chains
Use
- Energy storage
- plasma membrane of cells
- internal signalling (hormones)
Functional Groups
- Carboxyl group react with glycerol to form triglyceride
Proteins

Characteristics
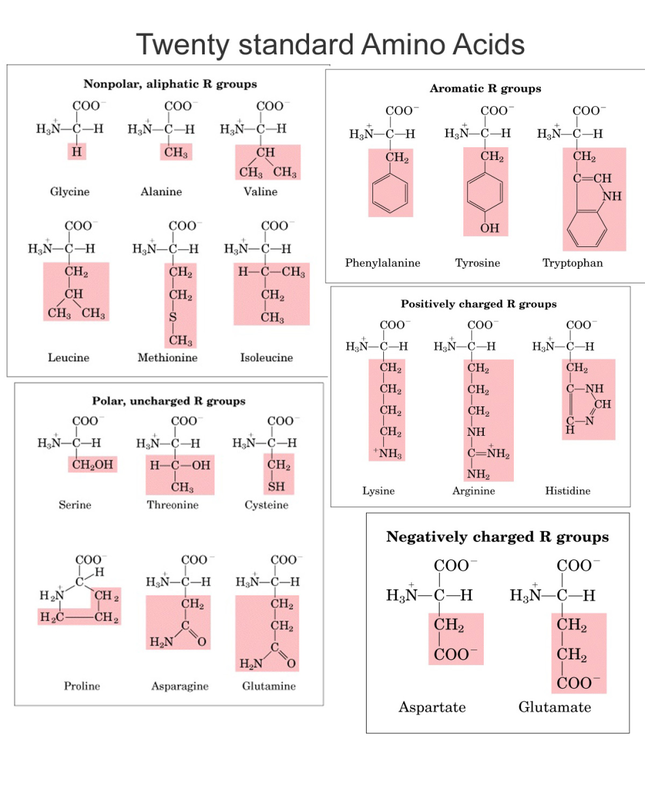
- Composed of amino acids bound by a peptide bond.
- There are 20 amino acids in the human body, which make up all the proteins in our bodies. (chart below)
Use
- Enzymes, which act as a catalyst in chemical reactions to speed them up
- Structure (cytoskeleton )
Functional Groups
- Amino and carboxyl groups
- Composed of amino acids bound by a peptide bond.
- There are 20 amino acids in the human body, which make up all the proteins in our bodies. (chart below)
Use
- Enzymes, which act as a catalyst in chemical reactions to speed them up
- Structure (cytoskeleton )
Functional Groups
- Amino and carboxyl groups
Nucleic Acids
Characteristics
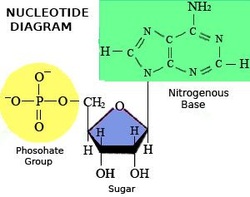
- Building block is a nucleotide
- For DNA the nucleotide is a nitrogen base and five carbon sugar and a phosphate group
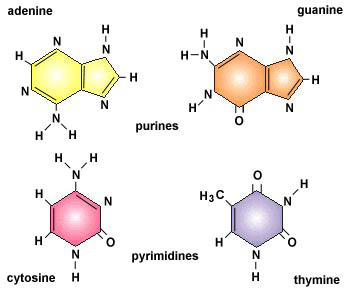
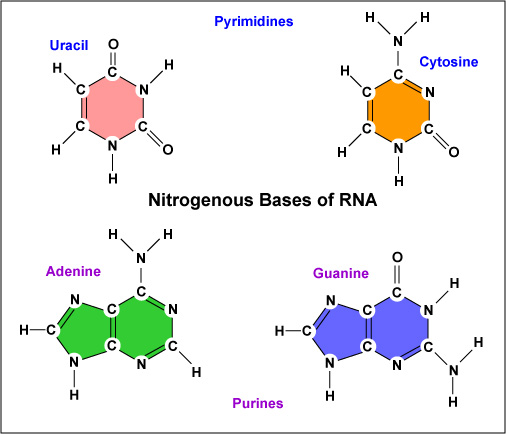
- There are five nitrogen bases used in the human body, in DNA and RNA. They are Adenine, Thymine, Guanine, Cytosine and Urasile
Use
- DNA (genetic material), RNA (making of proteins and flow of genetic info), ATP (energy transfer)
Functional Groups
- Hydroxyl groups and carbonyl groups
- Building block is a nucleotide
- For DNA the nucleotide is a nitrogen base and five carbon sugar and a phosphate group
- There are five nitrogen bases used in the human body, in DNA and RNA. They are Adenine, Thymine, Guanine, Cytosine and Urasile
Use
- DNA (genetic material), RNA (making of proteins and flow of genetic info), ATP (energy transfer)
Functional Groups
- Hydroxyl groups and carbonyl groups
assessment tools
| building_biochemical_molecules_handout.docx | |
| File Size: | 623 kb |
| File Type: | docx |
| mingle_to_music-_biological_compounds.docx | |
| File Size: | 13 kb |
| File Type: | docx |
Here is a lab written by James Dauray that examines ways of identifying biological compounds in food.
| biologically_important_molecules_chemical_tests_lab.docx | |
| File Size: | 145 kb |
| File Type: | docx |
Here is an article that discusses NYC ban on trans fats in restaurant foods. You could have the students read the article and discuss the problems with trans fats, compare then to saturated and unsaturated fats, and discuss how these things affect your body. This activity could also be done as a jigsaw, where one groups of students researches trans fats, another saturated, another unsaturated. The students would be expected to build a model to represent the type of fat they are talking about, point out the characteristics of the fat and discuss the implications to the body.